What is Aluminum foam?
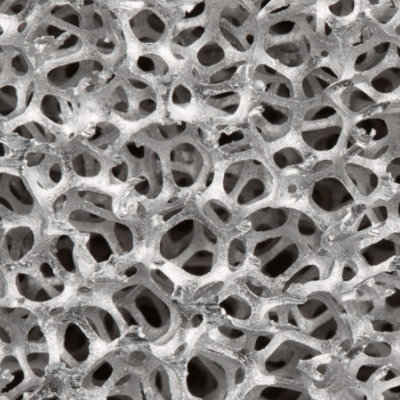
Aluminum foam is a rigid, three-dimensional network of interconnected aluminum ligaments that form cells with open pores. Duocel® Aluminum Foam contains completely solid ligaments (as opposed to hollow ligaments) and its properties are fully customizable, which allows its performance characteristics to be optimized for a broad range of applications.
Aluminum is ERG’s most popular Duocel® foam and is commonly used in applications that benefit from a lightweight structure, very high surface area, and from the nature-mimicking morphology of the rigid cellular structure.
How is Aluminum Foam used and what are its advantages?
Aluminum foam, also known as foamed aluminum, is used in a wide array of applications and industries. Below are just a few of the many examples and their benefits in each. For more examples and information, please refer to the Applications section.
- Mechanical Applications (e.g., structural components, impact/energy absorbers)
- High strength-to-weight
- Customizable strength
- Isotropic compression
- Thermal Applications (e.g., heat exchangers)
- High surface area per unit volume
- Creates turbulence in flowing fluids which minimizes boundary layers and promotes mixing to enhance thermal performance
- Custom conformal shapes/geometries to optimize size and weight
- Filtration and Media Separation (e.g., air-oil separators, flame arrestors, baffles)
- Tortuous path for media/debris to pass through
How is Aluminum Foam specified and customized?
Duocel® Aluminum Foam is defined by the following properties:|
Property |
Standard Option(s) |
Other Option(s) (Available on a limited basis or by special order) |
|
Aluminum Alloy |
6101 |
Other aluminum alloys (e.g., 1XXX, 6XXX, and 7XXX series, A356, etc.) |
|
Pores Per Inch (PPI) |
5, 10, 20, 40 |
N/A |
|
Relative Density |
4-12% |
Relative densities less than 4% and greater than 12% |
|
Compression Factor |
Uncompressed, 2X, 3X, 4X, 5X (up to approximately 50% relative density) |
6X+ depending on starting relative density and starting size (up to approximately 70% relative density) |
|
Heat Treatment Condition |
T6 |
F, O, T4 |
Our foams can be further customized by the following options:
Coating:
Surface coatings can be applied to enhance or augment the foam’s properties and protect against corrosion. Examples of a few coatings:
- Anodize (e.g. MIL-A-8625)
- Chemical Conversion (e.g. MIL-DTL-5541)
- Plating (e.g. gold, nickel, Teflon)
Joining:
Duocel® aluminum foam can be joined to other components. Examples of a few methods:
- Brazing (vacuum and dip)
- Adhesive Bonding
- Mechanical Fastening
- Interference and Thermal Fit
- Soldering
Example specification for an energy absorber application:
40 PPI, 8-10% relative density,
Processed from 6101 alloy,
Heat treated to T6 condition
What are Aluminum Foams physical characteristics?
Duocel® aluminum foam has many of the same characteristics as the parent aluminum alloy. Its open celled structure, however, does give foamed aluminium unique and customizable characteristics, such as compression strength and surface area. Please refer to the Technical and Applications sections for further details.
Example Physical Characteristics of Duocel® Aluminum Foam (6101-T6, 8% Relative Density):
| Compression Strength | 367 psi | (2.53 MPa) |
| Tensile Strength* | 180 psi | (1.24 MPa) |
| Shear Strength* | 190 psi | (1.31 MPa) |
| Modulus of Elasticity (Compression)* | 15 × 103 psi | (103.08 MPa) |
| Modulus of Elasticity (Tension)* | 14.6 × 103 psi | (101.84 MPa) |
| Shear Modulus | 2.9 × 104 psi | (199.95 MPa) |
| Vickers Pyramid Number | 35 HV | |
| Specific Heat | .214 BTU/lb-°F | (.895 J/g-C) |
| Bulk Thermal Conductivity | 3.4 BTU/ft-hr-F | (5.8 W/m-C) |
| Coefficient of Thermal Expansion (0-100°C) | 13.1 × 10-6 in/in–F | (23.58 × 10-6 m/m–C) |
| Bulk Resistivity | 2.84 × 10-5 ohm – in | (7.2 × 10-5 ohm – cm) |
| Melting Point | 1220°F | (660°C) |
What are the size limitations?
Duocel® aluminum foam is typically made in starting blocks up to approximately 16in x 16in x 4in (406mm x 406mm x 102mm) in size. Other sizes are available. Larger parts can be made by combining multiple pieces or by creating a custom size starting block.
There are also limits to how small or thin a part feature can be because cutting the open celled structure too small/thin results in no connected ligaments. The below table contains the standard minimum dimensions of uncompressed foam. Smaller dimensions and features are possible – please contact us to determine feasibility for your requirements.
|
PPI |
Standard Minimum Dimension |
|
5 |
0.35in (8.9mm) |
|
10 |
0.25in (6.4mm) |
|
20 |
0.20in (5.1mm) |
|
40 |
0.15in (3.8mm) |
Can it be fabricated to my custom shape/geometry?
Yes. Much like solid aluminum, Duocel® aluminum foam can be machined to your unique shape and dimensional requirements. Extra care must be taken for very small and tight-tolerance part features that can be impractical to manufacture or inspect (e.g., small holes, sharp edges or small radii, precise angles, etc.).
Can it be added as a part in an assembly?
Yes. Duocel® aluminum foam can be integrated into an assembly or system by joining (such as adhesive bonding and brazing), interference and thermal fits, and mechanical fastening. Foamed aluminum can make up an entire part, or just a small piece of a large system, such as a battery box with a foam flame arrestor.
How do I purchase samples of Duocel® Aluminum Foam?
Please visit an appropriate distributor, or contact us directly to discuss your application and request samples. In the United States, you may place your order online with a credit card at duocelfoam.com.
Characteristics:
- Lightweight
- High strength-to-weight ratio
- Customizable Strength
- High surface area-to-volume
Properties:
- Alloys: 6101 (standard), 1XXX, 6XXX, 7XXX
- PPI: 5, 10, 20, 40
- Relative Density: 4-12% (standard)
- Compression: Uncompressed to 5X (standard)
- Heat Treat Condition: T6 (standard), F, O, T4
- Optional:
- Coatings
- Joining/Assembly
All of our materials are proudly 100% Made in the USA.
